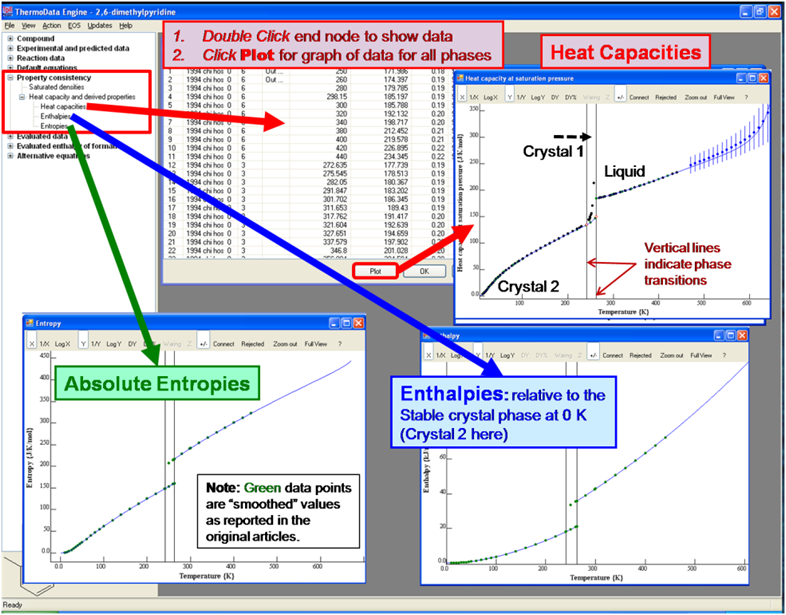
Navigation Tree: Property Consistency: Heat Capacities and Derived Functions
Absolute entropies and enthalpies (relative to T = 0 K) for condensed phases are evaluated by TDE based on experimental heat-capacity studies extending to low temperatures (T < 25 K). If multiple studies have been reported for a particular compound, TDE selects the best single data set for integration, and employs only the phase-transition properties from that source in the evaluations.Reports of experimental heat capacity measurements to low temperatures often include tables of smoothed thermodynamic functions. These derived properties are captured in TDE-SOURCE and are shown as green data points in the comparison plots below. The blue curves represent the property values evaluated by TDE. The plots of the enthalpies and entropies demonstrate the consistency between the smoothed values reported in the original articles (green) and the integrations by TDE (blue).
Evaluated and experimental data for all phases are shown on a single plot for each property.