PPDS12: For Hvap for the liquid-gas phase boundary
Hvap/(0.001×R ×Tc) = a1 × t1/3 + a2 × t2/3 + a3 × t + a4 × t2 + a5 × t6
where t = 1 - T/Tc, Tc is the critical temperature, and R is the gas constant.
Evaluation Results:
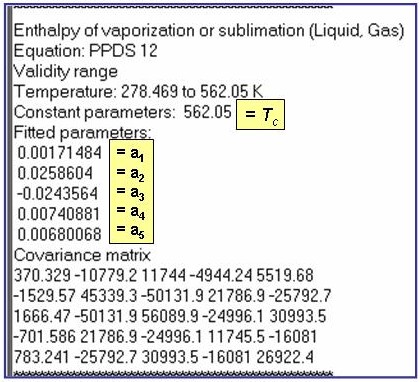
The example is for fitted enthalpies of vaporization for benzene.